The TimeWarp Protocol:
A Systems Approach to
Targeting Aging Biology
Inside the TimeWarp Longevity Optimization Engine
240
compounds evaluated
162
pathway–aging interactions mapped
~40K
intervention interactions evaluated
Based on more than 1,000 peer‑reviewed studies on human aging.
Most supplements target one ingredient or one outcome.
Aging does not work that way.
Aging is driven by interconnected biological systems. Cellular signaling networks interact, amplify, and influence multiple aging processes at once.
TimeWarp was designed using a systems engineering approach to longevity.
Instead of optimizing single ingredients, we optimized the entire protocol across multiple biological layers.
The result is a formula designed to influence the core biology of aging while remaining practical for daily use.
Engineered for Impact:
9 Design Principles
We engineered the formula using a proprietary optimization model guided by nine key factors:
Aging Pathways
Hallmarks of Aging
Mechanistic Impact
Scientific Evidence
Effective Dosing
Ingredient Synergy
Absorption Optimization
Protocol Usability
Long-Term Safety
Every capsule is the result of this multi-factor approach, designed to bring maximum impact in minimum daily effort.

We target the biological pathways that drive aging at the cellular level.

Our protocol is designed to support all 12 Hallmarks of Aging, the core processes linked to age-related decline.

We evaluate how cellular signaling networks interact so that interventions support multiple aging systems at once.

We prioritize compounds supported by human clinical research, not trends or emerging hype.
We upgrade selected compounds into clinically supported forms to improve uptake and systemic availability.

Each ingredient is included at levels shown to influence biological pathways in humans, not trace amounts for label appeal.

We combine compounds that influence complementary pathways for coordinated biological effects.

We design the protocol to fit within a realistic daily routine, not a 20+ capsule regimen.

Each ingredient is screened for tolerability and compatibility in long-term daily use.
TimeWarp Labs
Optimization Model Informational:
The TimeWarp Protocol was developed using mechanistic systems modeling and network-level analysis of the aging system.
Our research team built a multi-layer optimization engine that maps how molecular interventions influence the core mechanisms of aging.
The model evaluates longevity molecules across cellular pathways and the hallmarks of aging using evidence-weighted scoring.
We began by identifying the core biological pathways that regulate cellular aging, including metabolism, inflammation, repair, and mitochondrial function.
These upstream signaling networks served as the primary biological targets for candidate interventions.
Optimization Layer 1: AGING PATHWAYS
Pathway Modules and Precision Scoring
Pathway Modules (P₁–P₁₃) represent upstream cellular control networks through which interventions act.
We selected these modules because they:
Sit upstream of major aging phenotypes and influence downstream cascades
Can be modulated by evidence-backed, non-prescription interventions
Provide a structured bridge between compound mechanisms and system-level biology
Each pathway is further divided into specific regulatory submodules within that network. Compounds are scored only when they engage defined submodules with supporting evidence.
This prevents vague pathway claims and enforces mechanistic precision.
-
This pathway encompasses the most evolutionarily conserved longevity-relevant signaling axes. AMPK, mTORC1/2, insulin/IGF-1, and the sirtuin network collectively regulate the cell's investment in growth vs. maintenance, autophagy induction, stress resistance, and metabolic flexibility, making P1 the highest-weight pathway in this model.
SUBMODULES
Core Submodules:
AMPK energy sensing (AMP:ATP ratio detection; activation of catabolic programs)
mTORC1 nutrient and growth factor signaling (anabolic/proteostatic gatekeeper)
mTORC2 / PKB-Akt signaling axis (metabolic insulin sensitivity; FoxO phosphorylation)
Insulin/IGF-1 signaling control (IIS pathway; PI3K-PDK1 downstream)
NAD⁺ metabolism and sirtuin axis (SIRT1–7 as metabolic/epigenetic/mitochondrial effectors) [HOME: NAD salvage]
Important Submodules:
FOXO transcription factor programs (stress resistance, autophagy, antioxidant expression)
PGC-1α as nutrient-responsive mitochondrial biogenesis gate (AMPK/SIRT1-activated)
GCN2 and integrated amino acid sensing (eIF2α phosphorylation under amino acid limitation)
Metabolic flexibility and substrate switching (glucose-to-fatty-acid oxidation capacity)
Ketone signaling and fasting-mimetic state (β-hydroxybutyrate as HDAC inhibitor and NLRP3 suppressor)
Peripheral Submodules:
Adipokine signaling inputs (leptin/adiponectin as systemic metabolic modulators)
PPAR nuclear receptor axis (PPARα/γ/δ as lipid-sensing metabolic integrators)
REDD1/BNIP3-mediated AMPK-independent low-energy sensing (hypoxic metabolic adjustment)
-
The cellular redox system functions as a master sensor of molecular damage and energetic stress. NRF2-mediated antioxidant transcription, glutathione and thioredoxin systems, and NADPH-regenerating pathways collectively maintain the reductive capacity that protects DNA, proteins, and lipids from oxidative damage, a primary driver of genomic instability and mitochondrial dysfunction.
SUBMODULES
Core Submodules:
NRF2/KEAP1 antioxidant transcription (master regulator of phase-II and ARE-driven defense)
Glutathione system (GSH synthesis via GCL/GSS, recycling via GR, GSH:GSSG ratio maintenance)
NADPH regeneration and pentose phosphate pathway reductive capacity (G6PD/6PGD axis)
Superoxide dismutase and catalase enzymatic defense (SOD1/SOD2/CAT primary ROS scavenging)
Important Submodules:
Thioredoxin and peroxiredoxin systems (Trx1/2, Prx1–6; peroxide and disulfide management)
Heat shock response (HSF1 activation; HSP90/HSP70/HSP27 proteotoxic stress management)
Electrophile and xenobiotic response pathways (AhR, Nrf2-ARE via electrophilic inducers)
p38 MAPK/JNK stress kinase signaling (damage-responsive kinase amplification of stress signals)
MAPK-Erk1/2 as survival/proliferation-stress crosstalk mediator
Peripheral Submodules:
Metal redox handling (labile iron pool/LIP, copper chaperones; Fenton reaction management)
ROS hormetic threshold tuning (calibrating beneficial low-level ROS signaling vs. damage)
Ferroptosis defense axis (GPX4-mediated lipid peroxide clearance; FSP1/CoQ10 as backup)
-
Proteostasis: the maintenance of a healthy, functional proteome, is maintained by three interlocking systems: the ubiquitin-proteasome system for short-lived and misfolded proteins, macroautophagy for bulk cytoplasmic content, and lysosomal pathways for selective cargo. Mitophagy-specific machinery is homed in P4.
SUBMODULES
Core Modules:
Macroautophagy initiation and flux (ULK1/ULK2, Beclin-1/VPS34, ATG cascade, LC3 lipidation)
Lysosomal function, biogenesis, and acidification (TFEB/TFE3 axis; V-ATPase integrity)
Ubiquitin-proteasome system capacity (20S/26S proteasome assembly; E1-E2-E3 cascade)
Chaperone-mediated autophagy (CMA; LAMP-2A receptor availability; HSPA8 substrate selectivity)
Important Submodules:
ER stress and integrated stress response (PERK-eIF2α-ATF4, ATF6, IRE1α-XBP1 branches)
Heat shock chaperone network (HSP70/HSP90/GRP78 protein folding capacity)
Aggrephagy and p62/SQSTM1 scaffold-mediated aggregate clearance
Selective autophagy receptor diversity (NDP52/optineurin/NBR1 as cargo receptors)
Proteostasis transcriptional programs (HSF1-mediated chaperone gene induction)
Proteasome cap regulatory dynamics (19S/11S subunit composition and activity)
Peripheral Submodules:
Lipophagy (lipid droplet selective turnover via PLIN-LC3 interaction)
Reticulophagy / ER-phagy (FAM134B/RTN3 axis; ER quality membrane turnover)
Secretory autophagy and unconventional protein secretion (IL-1β/HMGB1 bypass pathway)
Xenophagy (selective autophagy of intracellular pathogens; aging immune relevance)
-
Mitochondrial integrity is central to cellular energetics, apoptotic priming, and ROS management. Age-associated mitochondrial dysfunction encompasses biogenesis decline, impaired quality control, mtDNA mutation accumulation, and loss of dynamic fidelity, collectively contributing to five of the twelve aging hallmarks.
SUBMODULES
Core Submodules:
Mitophagy machinery: PINK1/Parkin pathway, BNIP3/BNIP3L, FUNDC1 receptor-mediated mitophagy [HOME]
Mitochondrial biogenesis execution (PGC-1α/NRF1/NRF2/TFAM transcriptional cascade)
Respiratory chain integrity (ETC complexes I–IV, ATP synthase, supercomplex assembly)
Mitochondrial membrane potential maintenance (ΔΨᵐ; proton gradient integrity)
Important Submodules:
Fusion/fission dynamics balance (OPA1/MFN1-2 fusion; DRP1/FIS1/MFF fission)
UPRᵐᵗ and mitochondrial proteostasis (LONP1 protease, ClpP, mtHSP70, HSP60 chaperones)
mtROS generation, signaling, and mitochondrial redox (O₂⁻/H₂O₂ from Complex I/III; SOD2)
mtDNA integrity and replication fidelity (Polγ/TFAM/Twinkle helicase; heteroplasmy dynamics)
Mitochondrial calcium handling and buffering capacity (MCU complex; matrix Ca²⁺ homeostasis)
Peripheral Submodules:
Mitochondrial permeability transition pore (mPTP) sensitivity and apoptotic priming threshold
Mitochondrial metabolite signaling efflux (succinate/α-KG/acetyl-CoA as epigenetic substrates)
Cristae remodeling and inner membrane architecture (OPA1 oligomers; ETC supercomplex stability)
-
Genomic stability is maintained through an array of repair systems each specialized for distinct lesion types. Aging is accompanied by declining repair fidelity, accumulation of unresolved lesions, replication stress, and a progressive shift toward error-prone repair pathways, collectively driving mutational burden and senescence induction.
SUBMODULES
Core Submodules:
DNA damage response signaling (ATM/ATR kinase cascade; CHK1/CHK2 effector activation)
Double-strand break repair (homologous recombination via RAD51/BRCA1/2; NHEJ via Ku70/80/DNA-PKcs)
Nucleotide excision repair (global-genome NER via XPC/DDB2; transcription-coupled NER via CSA/CSB)
Base excision repair and oxidative lesion repair (OGG1/APEX1/XRCC1/PARP1 core)
Important Submodules:
Mismatch repair (MSH2/MSH6/MLH1/PMS2 axis; replication error correction)
Replication stress response and stalled replication fork stabilization (RPA/RAD51/FANC proteins)
PARP1/2 signaling as repair coordination and NAD⁺ consumer (hyperactivation in aging)
Chromosomal stability checkpoints (spindle assembly checkpoint; SAC via BUB1/BUBR1/MAD2)
Fanconi anemia pathway and interstrand crosslink repair (FANCD2/FANCI monoubiquitination)
Age-associated NHEJ vs. HR balance shift (end-joining fidelity decline in senescent cells)
Peripheral Submodules:
Nucleotide pool balance and dNTP availability (RRM2/p53R2 regulation; dNTP:rNTP ratio)
Translesion synthesis polymerases (Polη/ι/κ bypass; mutagenic risk vs. fork rescue)
R-loop resolution and transcription-replication conflict (AQR/SETX helicases)
Repair factor expression regulation (downstream transcriptional control; avoid P7 overlap)
-
Telomere attrition represents one of the most cell-autonomous and quantifiable aging clocks in somatic tissues. Telomere maintenance involves not only the telomerase complex but a sophisticated shelterin architecture, DNA damage surveillance at uncapped ends, and heterochromatin maintenance across the telomeric repeat region.
SUBMODULES
Core Submodules:
Telomerase regulation (TERT catalytic subunit; TERC template RNA; dyskerin/TCAB1 holoenzyme)
Shelterin complex integrity and end protection (TRF1/TRF2/POT1/TPP1/RAP1/TIN2 hexameric complex)
Telomere damage response signaling (uncapping-induced TIFs; ATM/ATR activation at telomeres)
Important Submodules:
Replicative senescence checkpoints triggered by critically short telomeres (p21/p16 induction)
Telomere chromatin state maintenance (H3K9me3/HP1α heterochromatin; ATRX/DAXX deposition)
T-loop structural dynamics and G-quadruplex secondary structure resolution (FANCJ/BLM/RHAU/DHX36 helicases)
TERRA (telomere repeat-containing RNA) transcription and R-loop regulation at telomeres
Peripheral Submodules:
Telomere position effect on proximal gene expression (TPE and TPE-OLD via H3K9me3 spreading)
Alternative lengthening of telomeres pathway (ALT; PML bodies; ATRX-DAXX loss; BLM-mediated)
Oxidative susceptibility of telomeric guanine residues (8-oxo-dG preferential accumulation)
-
Epigenetic alterations constitute one of the strongest predictors of biological age, with DNA methylation clocks (Horvath, 2013) achieving remarkable precision. Age-associated epigenetic drift spans methylation patterns, histone marks, chromatin accessibility, and 3D nuclear architecture, collectively disrupting transcriptional fidelity and cell identity maintenance.
SUBMODULES
Core Submodules:
DNA methylation maintenance and age-associated drift control (DNMT1/DNMT3a/3b; TET1/2/3 demethylases)
Histone acetylation/deacetylation balance (HAT/KAT complex activity; HDAC class I/II/III regulation)
Chromatin accessibility remodeling (SWI/SNF/BAF, ISWI, NuRD ATP-dependent remodeling complexes)
Polycomb/Trithorax antagonism (PRC2 H3K27me3 silencing vs. TrxG/MLL H3K4me3 activation)
Important Submodules:
Histone methylation and demethylation dynamics (EZH2/KDM family; H3K4me3/H3K27me3/H3K9me3)
Transcriptional program stability and cell identity maintenance (enhancer-promoter connectivity fidelity)
Pioneer transcription factor activity and chromatin reprogramming capacity (FOXA/GATA/OCT4-class)
RNA processing and splicing regulation as epigenetic interface [HOME] (co-transcriptional splicing fidelity)
3D chromatin organization and TAD boundary integrity (CTCF/cohesin loops; lamin B1-mediated architecture)
Non-coding RNA regulation (lncRNA enhancer RNA; epigenetic silencing via HOTAIR/XIST-class)
Peripheral Submodules:
Transposon and repeat element repression (LINE-1/HERV-K silencing via H3K9me3/DNMT3/PIWI)
Histone variant incorporation and turnover (H3.3/H2A.Z dynamics; HIRA/ATRX-mediated)
DNA damage-induced chromatin changes (γH2AX spreading; PARP-mediated chromatin relaxation)
Epitranscriptomic modifications (m⁶A/pseudouridine as mRNA stability and translation regulators)
-
Cellular senescence represents an antagonistic hallmark: initially a tumor-suppressive and wound-healing mechanism, chronic senescent cell accumulation drives tissue dysfunction through SASP-mediated paracrine damage, immune evasion, and bystander senescence propagation. This pathway is the direct mechanistic target of the TimeWarp monthly senolytic protocol.
SUBMODULES
Core Submodules:
Senescence induction and stable cell-cycle arrest (p16ᶤᴺᴴᵃ/CDK4-6/Rb; p53/p21/CIP1 axis)
NF-κB canonical/non-canonical inflammatory transcriptional activation (IKKβ/IKKα/NEMO complex)
cGAS-STING cytosolic DNA sensing (mtDNA/cytoplasmic chromatin fragments as innate immune activators)
SASP program generation: cytokine/chemokine/protease production [HOME] (IL-6/IL-8/MMP3/GROa/PAI-1)
Important Submodules:
NLRP3 and related inflammasome platform assembly and activation (IL-1β/IL-18 processing)
JAK1/2-STAT1/3/5 inflammatory signaling axis (cytokine receptor downstream in SASP context)
Complement system activation as aging-associated tissue amplifier (C1q/C3/MAC deposition)
mTORC1-driven translational enhancement of SASP (4EBP1/S6K1 axis; cap-dependent SASP mRNA translation)
Senescence immune surveillance and clearance failure (NK cell/macrophage senescent cell recognition)
Peripheral Submodules:
Bystander senescence induction via paracrine SASP signals (local tissue propagation; gap junctions)
Resolution signaling and counter-regulatory balance (SPMs/lipoxins; IL-10/TGF-β as brakes)
Cytokine receptor desensitization and inflammatory tolerance (signal attenuation mechanisms)
Neutrophil extracellular trap (NET) formation in aged tissue (NETosis as inflammaging amplifier)
-
Stem cell exhaustion represents the integrative consequence of upstream damage accumulating in long-lived progenitor pools. Signaling pathways governing self-renewal, quiescence, and niche communication are progressively corrupted by SASP exposure, epigenetic drift, and metabolic dysregulation, collectively impairing tissue renewal and regenerative capacity.
SUBMODULES
Core Submodules:
Wnt/β-catenin signaling control of stem cell fate, self-renewal, and symmetric division
Notch signaling for progenitor specification and tissue homeostasis (Notch1/2/3; DLL/Jagged ligands)
TGF-β/BMP signaling gate (regeneration vs. fibrosis balance; Smad2/3 and Smad1/5/9 axes)
Important Submodules:
Hedgehog signaling in tissue-specific stem cell niches (Gli1/2 as effectors in hair/muscle/intestine)
HIF-1α/HIF-2α hypoxic niche maintenance and stem cell quiescence regulation
mTOR-mediated metabolic control of quiescence vs. activation (HSC exhaustion via mTORC1 hyperactivation)
Growth factor receptor signaling for repair (EGFR/FGFR1-4/cMet as proliferative drivers)
Niche-derived retention and mobilization signals (SCF/CXCL12/Ang-1/VCAM-1 niche architecture)
Peripheral Submodules:
Lineage commitment tuning and epigenetic poising (tissue-specific differentiation fidelity)
Asymmetric vs. symmetric division balance (aging-associated shift toward symmetric; exhaustion risk)
Senescence-mediated niche disruption (paracrine SASP from P8; context-dependent scoring)
ECM remodeling as niche mechanical signal (interface with P10; scored peripherally here)
-
The extracellular matrix serves as both a structural scaffold and a signaling environment for tissue-resident cells. Age-associated matrix stiffening, collagen crosslinking, and disrupted MMP/TIMP balance progressively alter mechanosensing, impair stem cell niche function, and create a pro-fibrotic and pro-inflammatory tissue milieu.
SUBMODULES
Core Submodules:
ECM remodeling balance (MMP/TIMP ratio; collagenase/gelatinase/stromelysin activity control)
Mechanotransduction signaling via YAP/TAZ nuclear translocation and Hippo pathway (LATS1/2)
Matrix stiffness sensing and fibrotic reprogramming (TGF-β-driven myofibroblast differentiation)
Important Submodules:
Integrin-FAK-Src signaling complex and adhesion mechanics (focal adhesion kinase/paxillin)
Collagen crosslinking, glycation, and AGE-driven matrix stiffening (LOX/transglutaminase; RAGE signaling)
Basement membrane integrity (laminin/perlecan/collagen IV/nidogen assembly and turnover)
Fibronectin remodeling and RGD-motif-mediated cell-matrix communication
Peripheral Submodules:
Glycocalyx composition and microvascular interface mechanics (heparan sulfate proteoglycans)
Proteoglycan turnover (versican/aggrecan/biglycan remodeling; hyaluronan homeostasis)
Tissue oxygenation via matrix tortuosity and diffusion constraints (context-dependent)
Mechanosensitive ion channels as upstream transducers (Piezo1/Piezo2; TRPV4)
-
Beyond cell-autonomous mechanisms, aging is shaped by circulating factors, extracellular vesicles, and neuroendocrine signals that coordinate tissue states across the organism. The progressive disruption of positive systemic signals and accumulation of negative ones, including SASP components, defines the altered intercellular communication hallmark.
SUBMODULES
Core Submodules:
Endocrine and paracrine signaling coordination (circulating hormone/growth factor axis)
Extracellular vesicle and exosome cargo signaling [HOME] (miRNA/protein/lipid payload transfer)
Systemic propagation of damage and resilience signals (parabiosis-relevant circulating factors)
Important Submodules:
SASP propagation and circulating cytokine/chemokine systemic impact [HOME] (non-generation aspect)
Circulating rejuvenation factors (GDF11/Klotho/TIMP2/GPC1; age-associated decline)
Neuroendocrine coordination via hypothalamic-pituitary axis (GnRH/GH/IGF-1 decline; hypothalamic inflammation)
Immune-to-tissue signaling crosstalk and immune editing of aged tissues (inflammaging mediators)
Senolytic immune surveillance and clearance failure of pro-aging senescent cells (NK/T-cell dysfunction)
Peripheral Submodules:
Circulating miRNA species as systemic regulators (subset of EV biology; independent evidence required)
Hormone receptor sensitivity shifts and downstream signaling efficacy changes (receptor desensitization)
Platelet-derived aging factors (PF4/RANTES age-associated release as systemic inhibitors)
Bone marrow niche-derived systemic signals (TPO/SCF as hematopoietic aging regulators)
-
The circadian clock is now recognized as a master upstream regulator of metabolic, immune, and repair processes across virtually every tissue. Age-associated circadian dampening, reduced BMAL1 amplitude, increased CRY phosphorylation, and desynchrony between central and peripheral oscillators, accelerates epigenetic aging and metabolic dysregulation (Kondratov et al., 2006; Thaiss et al., 2016).
SUBMODULES
Core Submodules:
Core oscillatory clock machinery integrity (CLOCK/BMAL1 positive arm; PER1/2-CRY1/2 negative feedback)
Circadian gating of metabolic control (NAMPT/NAD⁺ oscillation; clock-driven lipid/glucose metabolism)
Circadian regulation of immune responsiveness and inflammatory amplitude (NF-κB clock crosstalk)
Important Submodules:
Circadian control of autophagy and lysosomal function timing (BMAL1-TFEB axis)
Circadian influence on DNA repair capacity and timing (BMAL1-XPA-NER pathway; repair rhythmicity)
Circadian regulation of mitochondrial dynamics, OXPHOS rhythmicity, and ROS management
Peripheral clock synchronization (liver/gut/muscle/adipose tissue oscillators vs. SCN)
Melatonin axis as systemic circadian entrainer, antioxidant, and mitochondrial protector
Peripheral Submodules:
Sleep architecture coupling and restorative processes (downstream of clock; NREM slow-wave depth)
Feeding-time alignment and chrono-nutrition effects (chrono-restriction; TRF benefits)
Temperature compensation of clock period (Q10 near-1.0 mechanisms; CK1ε/δ period tuning)
Circadian gating of senescence and SASP amplitude (BMAL1 loss-accelerated SASP in fibroblasts)
-
The intestinal microbiome constitutes a dynamic signaling interface with host immunity, metabolism, and epigenetics. Age-associated dysbiosis — characterized by reduced diversity, Firmicutes/Bacteroidetes ratio shifts, and barrier disruption — contributes to systemic inflammation, metabolic dysfunction, and accelerated biological aging (Claesson et al., 2012; Thevaranjan et al., 2017).
SUBMODULES
Core Submodules:
Intestinal barrier integrity and permeability signaling (tight junctions: claudin/occludin/ZO-1)
Microbial metabolite-to-host signaling (SCFAs: butyrate/propionate/acetate; urolithins; equol)
Immune modulation via microbiome-host pattern recognition (TLR/NOD/CLR; innate-adaptive bridge)
Important Submodules:
Bile acid metabolism and microbial biotransformation (primary-to-secondary bile acid conversion; FXR/TGR5)
Microbiome influence on systemic inflammatory tone (LPS/endotoxemia; metabolic endotoxemia; TLR4 activation)
Microbiome effects on nutrient sensing inputs (butyrate-mediated AMPK activation; GPR41/43 SCFA signaling)
Tryptophan-derived microbial signaling (indoles/AhR axis; IDO1-kynurenine-NAD⁺ pathway)
Microbial diversity, ecological stability, and age-associated community composition shifts
Peripheral Submodules:
Circadian effects on microbiome composition and diurnal oscillation (home = P12; peripheral credit only)
Microbiome effects on epigenetic drift via metabolite supply (SAM/butyrate/folate availability)
Phage-bacteria dynamics and microbiome stability (virome-microbiome community interactions)
Microbiome effects on mitochondrial function (secondary bile acid/SCFA crosstalk; UCP2 regulation)
Mucosal IgA-mediated microbiome shaping and homeostatic immune tolerance
13 biological aging pathways modeled, 200+ molecular mechanisms mapped
Optimization Layer 2: HALLMARKS OF AGING
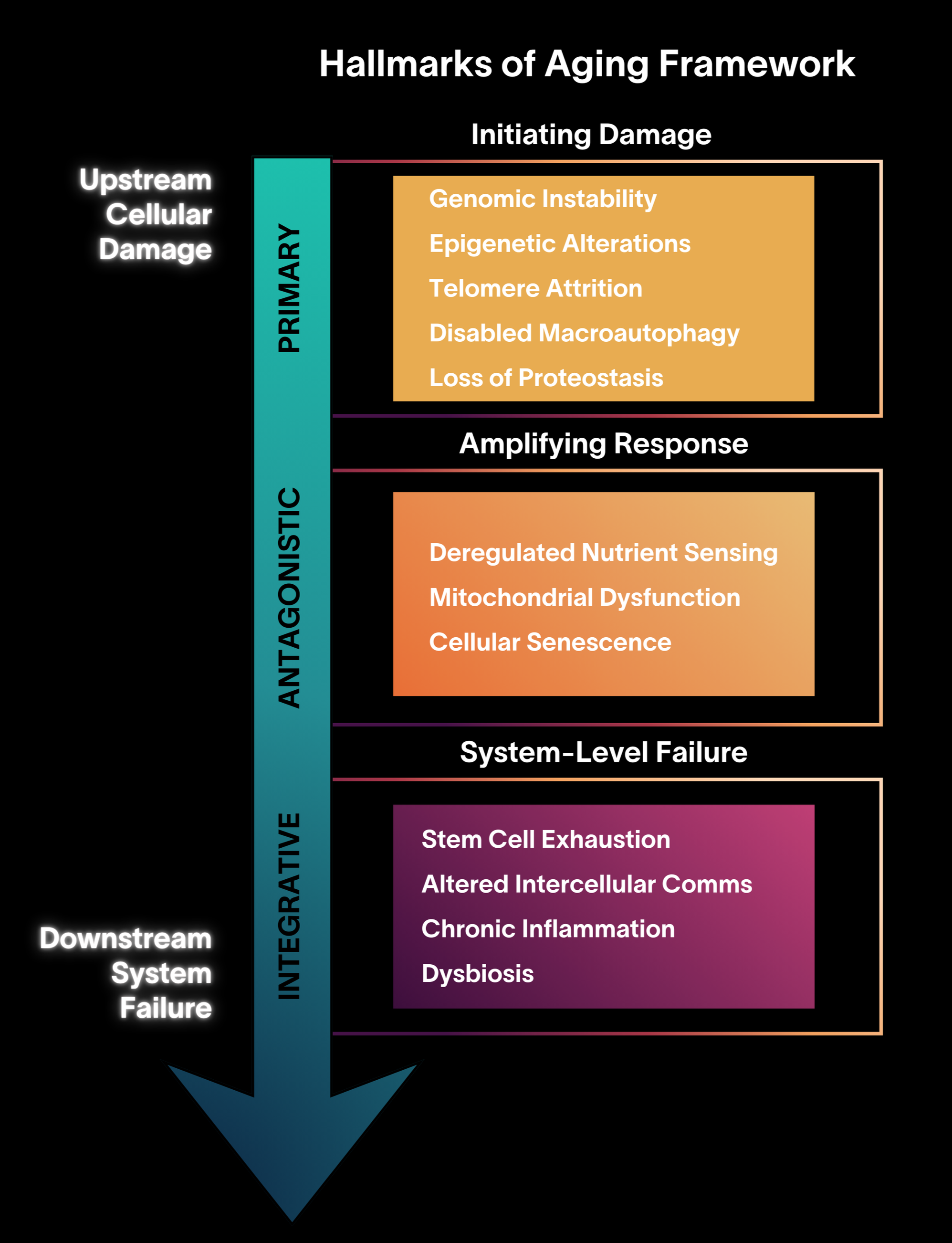
The second layer of the model is the 12 Hallmarks of Aging.
The hallmarks describe the biological consequences of aging.
Upstream cellular pathways drive these changes.
The hallmarks allow us to measure the downstream effects.
Our approach focuses on influencing the pathways so their impact on the hallmarks improves.
Not all hallmarks play the same role in aging.
Some represent upstream sources of cellular damage, while others reflect downstream consequences that emerge over time.
Our optimization model accounts for this structure. By focusing on upstream biological drivers, interventions can influence multiple aging processes at once.
The diagram illustrates how the 12 hallmarks interact across this upstream-to-downstream spectrum and how pathway interventions propagate through the system.
12 hallmarks of aging integrated
+ 13 Aging Pathways
+ 162 Aging Pathway Submodule
Relationships mapped
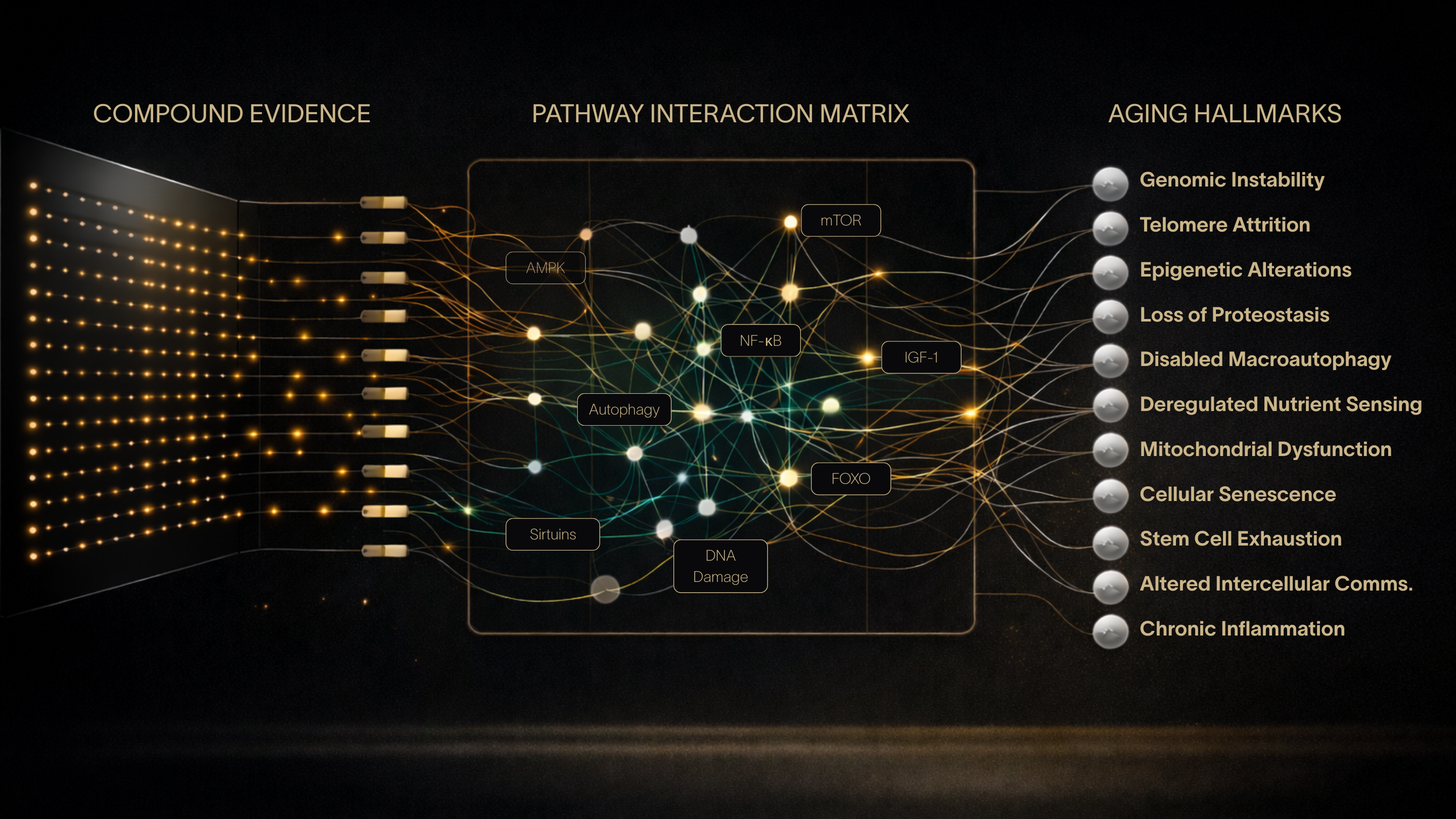
Optimization Layer 3: MECHANISMS
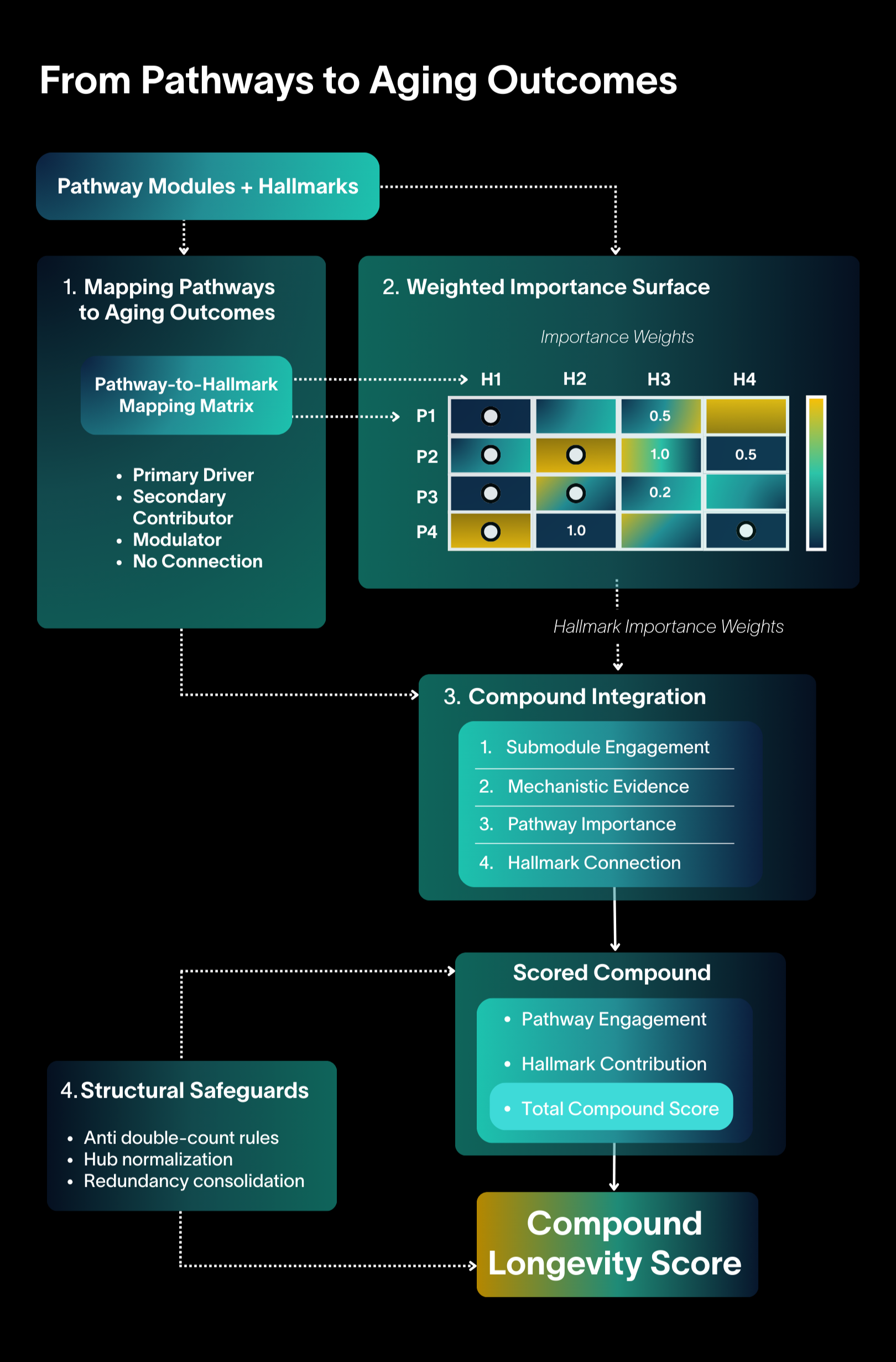
A weighted pathway-hallmark matrix evaluates mechanistic coverage to produce a structured compound score. This layer freezes the compound scoring and evaluationarchitecture.
-
Each pathway’s relationship to aging systems is graded as primary, secondary, modulator, or none. This prevents inflated credit.
-
Pathway importance, hallmark importance, and their connections are combined into a weighted importance surface. This defines how biological influence propagates through the system.
-
Compounds are evaluated by the specific submodules they engage and the strength of evidence. Engagement propagates through the weighted surface to produce a total score.
-
Anti double-count rules, hub normalization, and redundancy controls prevent distortion.
Hundreds of mechanistic pathway interactions evaluated
Optimization Layer 4: COMPOUND UNIVERSE
We mapped the full landscape of compounds studied for their potential to influence aging biology.
This library, which we call the Compound Universe, includes natural molecules, plant compounds, and nutraceutical ingredients supported by mechanistic research, preclinical studies, and human evidence.
-
Inclusion required:
Commercial availability in the US or EU
Peer-reviewed mechanistic support
Safety at commonly supplemented doses
Clear mappability to defined pathway submodules
Practical formulatability
Exclusions included:
Prescription-only agents
Research-only compounds
Molecules without sufficient mechanistic resolution
Compounds with regulatory or safety constraints
-
The final universe included 240 non-prescription compounds across 28 pharmacological classes.
Grouped by pharmacology, not pathway relevance, to avoid structural bias.
Family controls were applied to prevent redundancy:
Delivery-form variants share mechanistic records
Distinct relatives are scored independently
Structural clustering is flagged in rankings
Select prescription agents were retained only as mechanistic benchmarks to anchor scoring rigor.
Every compound was evaluated before any formulation decision was made.
We evaluated each compound using a structured evidence framework that connects mechanisms, pathways, and aging outcomes.
Rather than relying on individual studies, we assess how compounds influence aging biology as a system.
Each candidate is evaluated for:
• Pathway influence
• Hallmark-level impact
• Human clinical evidence
• Compatibility with other interventions
Optimization Layer 5: EVIDENCE MAPPING
The result is a ranked shortlist of longevity interventions with the strongest mechanistic influence on the drivers of aging.
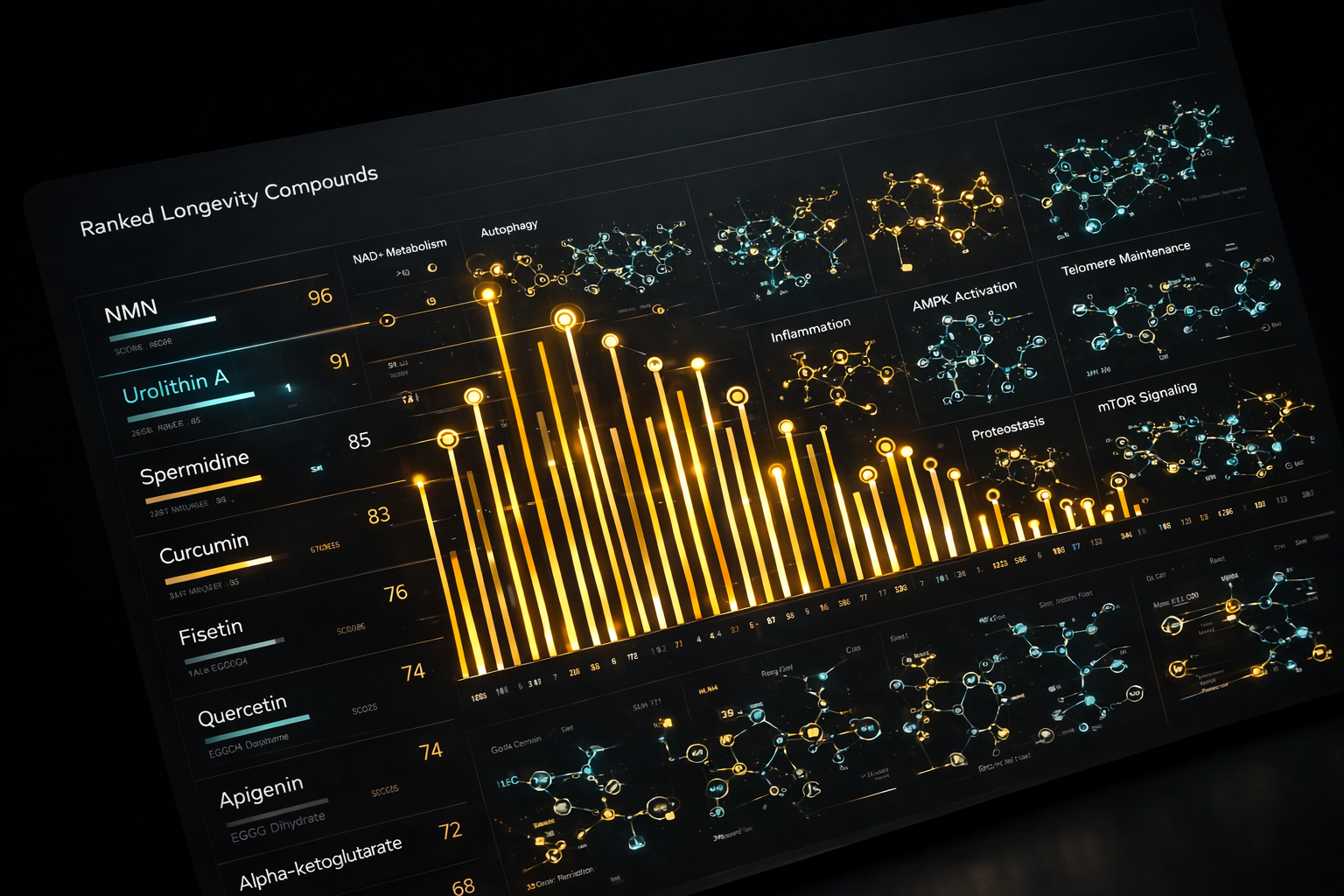
Filtering, Ranking and Scoring example for illustrative purposes only:
240 longevity molecules evaluated
~40K molecular interactions analyzed
Optimization Layer 6: COMPOUND INTERACTIONS
High-ranking compounds were then evaluated as a system, accounting for synergy, redundancy, and safety interactions.
Illustrative Image:
Synergistic Amplification
Some compounds influence complementary pathways and strengthen each other's effects when combined.
The model identifies interactions where multiple compounds produce stronger pathway activation than individual interventions.
Redundancy & Overlap
Compounds that act through nearly identical mechanisms are evaluated for redundancy.
When two candidates produce similar pathway effects, the model favors the compound with stronger evidence, broader pathway coverage, or better delivery characteristics.
Safety Interaction Screening
Potential compound interactions are also evaluated during this stage.
Candidates that introduce unfavorable interaction profiles, weak system contribution, or poor compatibility with the broader stack are removed before formulation design.
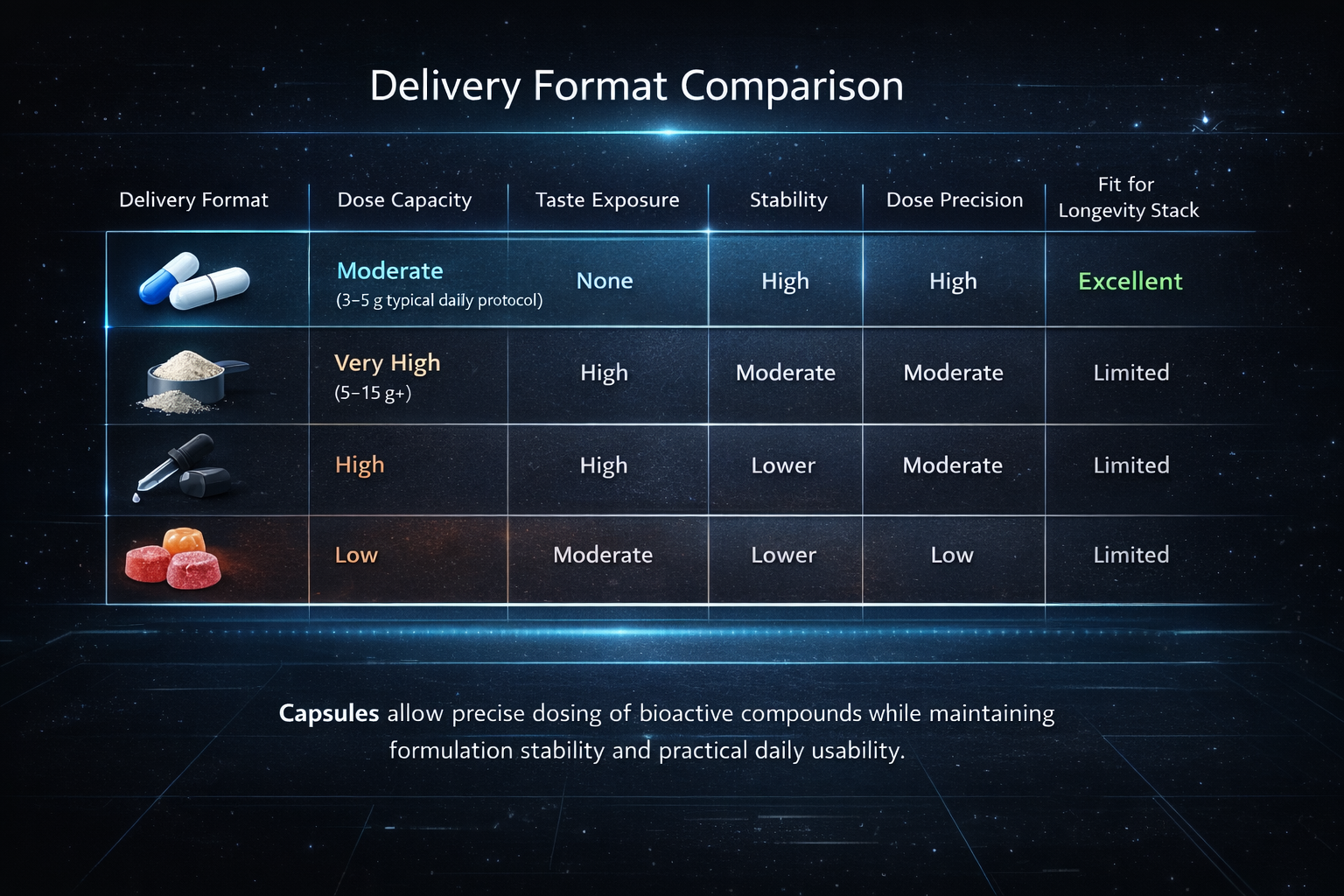
Delivery Format
Multiple delivery formats were evaluated during formulation design, including capsules, powders, liquids, and gummies.
Each format introduces different constraints related to dosing capacity, ingredient stability, taste masking, and daily usability.
Many longevity compounds require meaningful gram-level dosing and have naturally bitter or sulfur-containing flavor profiles that are difficult to mask in liquid or gummy systems.
Capsules provide a neutral delivery format that allows precise dosing while avoiding taste masking challenges and excessive sugar or flavoring systems.
Optimization Layer 7: FORMULATION & DELIVERY
Feasibility Filtering
Compounds that cannot be delivered within the defined protocol architecture are removed or replaced with more practical alternatives.
This ensures that every compound in the final formulation can be delivered at a meaningful biological level.
Dose Optimization
Each remaining compound is evaluated against evidence-based dosing ranges to ensure the protocol reflects meaningful biological exposure.
Bioavailability Engineering
For compounds that remain in the formulation, delivery formats may be optimized to improve absorption and cellular availability.
Enhanced ingredient forms can be used where necessary to ensure compounds reach their intended biological targets.
Delivery Architecture
The protocol was designed around six size-00 capsules per day, a format widely used in advanced nutraceutical formulations because it balances ingredient capacity, swallowability, and daily adherence.
This architecture allows the protocol to deliver meaningful research-level dosing while remaining practical for daily use.
The formulation must operate within real-world delivery limits such as capsule capacity, ingredient density, and daily usability.
These physical constraints define how much active material can realistically be delivered within a practical daily protocol.
The result is a formulation architecture that preserves the scientific integrity of the optimization model while translating it into a practical longevity protocol.
Optimization Layer 8: IMPLEMENTATION
Translating the optimized system into a practical longevity protocol
After compound selection, synergy optimization, and formulation engineering are completed, the final stage of the model evaluates practical factors that influence whether the protocol can be successfully used in the real world.
Even the most scientifically optimized compound stack must ultimately translate into a product that people can consistently integrate into their daily routine.
This stage ensures the final protocol balances scientific rigor, manufacturing feasibility, and long-term usability.
Cost & Manufacturing Feasibility
Some compounds with promising biological mechanisms may be impractical due to extreme cost, unstable supply chains, or manufacturing limitations.
The model evaluates ingredient availability, production scalability, and formulation feasibility to ensure the final protocol can be sustainably manufactured while maintaining high-quality ingredient standards.
Perceptible Benefits & Daily Experience
While the protocol is primarily designed around longevity pathways and aging mechanisms, certain compounds may also provide perceptible benefits such as improved energy, focus, or mood.
These effects can reinforce daily adherence by allowing users to experience immediate benefits while the deeper biological mechanisms operate over time.
Safety & Compatibility
Potential ingredient interactions and tolerability considerations are evaluated to ensure the compounds in the final protocol are compatible when used together.
This step ensures the final stack maintains a balanced formulation suitable for long-term use.
Protocol Architecture & Usability
To improve clarity and usability, the final protocol organizes compounds into functional groups that reflect their primary roles within the system.
These groups are visually differentiated and structured to help users understand how each component contributes to the overall longevity protocol.
This approach simplifies a complex multi-compound system into a format that is easier to follow consistently over time.
Result
The result is a longevity protocol that balances scientific optimization, formulation engineering, and real-world usability, translating complex aging research into a system that can be consistently applied in everyday life.